Table of Contents
- Introduction
- Battery cell C-rate
- Battery cell discharge characteristic
- Battery cell internal resistance circuit model
- Internal resistance calculation
- Open circuit voltage calculation
- Power loss calculation
- Conclusions
- References
Introduction
The internal resistance of a battery cell is a measure of the resistance to the flow of current within the cell. It is typically expressed in units of ohms (Ω).
Internal resistance can be thought of as a measure of the “quality” of a battery cell. A low internal resistance indicates that the battery cell is able to deliver a large current with minimal voltage drop, while a high internal resistance indicates that the battery cell is less able to deliver a large current and experiences a larger voltage drop.
Internal resistance can be affected by various factors, including: the type and composition of the electrodes, the temperature of the cell, and the state of charge of the cell. It can also vary depending on the discharge rate, with higher discharge rates typically resulting in a higher internal resistance.
Measuring the internal resistance of a battery cell can be useful for determining the performance of the cell and identifying any issues that may affect its performance.
For a lithium-ion battery cell, the internal resistance may be in the range of a few mΩ to a few hundred mΩ, depending on the cell type and design. For example, a high-performance lithium-ion cell designed for high-rate discharge applications may have an internal resistance of around 50 mΩ, while a lower-performance cell designed for low-rate discharge applications may have an internal resistance of around 200 mΩ.
For a lead-acid battery cell, the internal resistance may be in the range of a few hundred mΩ to a few thousand mΩ. For example, a deep-cycle lead-acid battery designed for use in an electric vehicle may have an internal resistance of around 500 mΩ, while a high-rate discharge lead-acid battery may have an internal resistance of around 1000 mΩ.
For a nickel-metal-hydride (NiMH) battery cell, the internal resistance may be in the range of a few hundred mΩ to a few thousand mΩ. For example, a high-capacity NiMH battery cell designed for use in an electric vehicle may have an internal resistance of around 1000 mΩ, while a high-rate discharge NiMH battery may have an internal resistance of around 2000 mΩ.
It is common for battery cell manufacturers to provide information on the internal resistance of their cells in their datasheets. This information can be useful for designers and engineers who are selecting batteries for a particular application, as it can help them to understand the performance characteristics of the cell and how it may behave under different conditions.
The internal resistance of a battery cell is typically provided as a static value, which reflects the resistance of the cell when it is at a relatively low discharge rate (usually around 0.2C or less). At higher discharge rates, the internal resistance of the cell may be higher.
In some cases, the datasheet may provide information on the internal resistance of the cell at different discharge rates, which can be useful for understanding the cell’s performance under a wider range of conditions.
If the internal resistance of the battery cell is not provided by the manufacturer, as we’ll see in this article, using the discharge characteristics of the battery cell, we can calculate the internal resistance of the battery cell, for a specific state of charge value.
Battery cell C-rate
The C-rate of a battery cell refers to the rate at which a battery is charged or discharged. It is expressed as a multiple of the cell’s capacity, with a C-rate of 1C indicating that the cell is being charged or discharged at a rate equal to its capacity, a C-rate of 2C indicating that it is being charged or discharged at twice its capacity, and so on.
For example, if a battery cell has a capacity of 1000 mAh and is being discharged at a rate of 2C, it is being discharged at a rate of 2000 mA (2 x 1000 mA).
In the table below you can find a detailed explanation on different C-rate for a battery cell with the capacity of 3200 mAh.
| C-rate | Discharge current | Discharge time (complete discharge) |
| 0.2C | 0.2 · 3200 mAh = 640 mA | 3200 mAh / 640 mA = 5 hours |
| 0.5C | 0.5 · 3200 mAh = 1600 mA | 3200 mAh / 1600 mA = 2 hours |
| 1C | 1 · 3200 mAh = 3200 mA | 3200 mAh / 3200 mA = 1 hour |
| 2C | 2 · 3200 mAh = 6400 mA | 3200 mAh / 6400 mA = 0.5 hours |
The C-rate of a battery cell can be an important factor in its performance and lifespan. In general, higher C-rates can result in faster charge and discharge times, but may also lead to a reduction in the overall lifespan of the cell due to increased stress on the electrodes and other components.
It is important to note that the C-rate of a battery cell can vary depending on the cell’s design and chemistry, as well as the temperature and state of charge of the cell. Some cells may be able to sustain higher C-rates without significant degradation, while others may be more sensitive to high C-rates.
Battery cell discharge characteristic
The discharge characteristic of a battery cell refers to the way in which the cell’s voltage and capacity change as it is discharged. The discharge characteristic of a cell can be an important factor in its performance and can vary depending on the type of cell, its design and chemistry, and the conditions under which it is being discharged.
In general, the voltage of a battery cell decreases as it is discharged, and the rate at which the voltage decreases can depend on the load being placed on the cell and the state of charge of the cell. For example, a cell that is being discharged at a high rate (i.e., a high C-rate) may experience a more rapid voltage drop than a cell that is being discharged at a low rate.
The capacity of a battery cell also decreases as it is discharged, and the rate at which the capacity decreases can depend on the cell’s design and chemistry, as well as the discharge rate and temperature. The capacity of a cell is typically measured in milliampere-hours (mAh), and it represents the amount of energy that the cell can deliver over a given period of time.
For example, consider a lithium-ion battery cell with a capacity of 1000 mAh. If the cell is discharged at a rate of 1C (i.e., 1000 mA), it will be completely discharged in approximately one hour. If the discharge rate is increased to 2C (i.e., 2000 mA), the cell will be completely discharged in approximately half an hour.
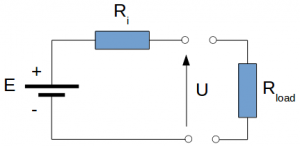
Battery cell internal resistance circuit model
The battery cell circuit model is a mathematical model that represents the behaviour of a battery cell in an electrical circuit. It is based on the cell’s internal resistance Ri [mΩ], open circuit voltage E [V], and terminal voltage U [V].
The internal resistance of a battery cell Ri [mΩ] is a measure of the cell’s resistance to the flow of current. It is caused by various factors, such as the cell’s electrode material, the thickness of the electrodes, and the ionic conductivity of the electrolyte. The internal resistance of a cell can affect its performance and efficiency, and it is typically higher at higher current densities and lower temperatures.
The open circuit voltage E [V] of a battery cell is the voltage of the cell when it is not connected to any external load. It represents the cell’s electrochemical potential and is influenced by various factors, such as the cell’s state of charge, temperature, and age.
The terminal voltage U [V] of a battery cell is the voltage measured at the cell’s terminals when it is connected to an external load. It is equal to the open circuit voltage minus the voltage drop caused by the cell’s internal resistance and the external load.
U = E – I·Ri (eq. 1)
The electrical current I [A] is the current passing through the internal resistance when there is a load connected to the battery cell.
The battery cell circuit model can be used to predict the cell’s voltage, current, and state of charge under different conditions, such as different load currents and temperatures. It can be useful for applications such as battery management systems, where a fast and accurate representation of the cell’s behaviour is desired.
Internal resistance calculation
If we have the discharge characteristics of a battery cell, for different values of the C-rate, we can calculate the internal resistance of the battery cell at a specific state of charge (SoC) [1].
As an example we are going to use the Panasonic NCR18650B lithium-ion battery cell which has a nominal capacity of 3200 mA and the discharge characteristics as in the image below [2].
We aim to calculate the internal resistance of the cell at approximatively 47 % state of charge (SoC).
Step 1. Calculate the discharge capacity of the battery cell for 47 % SoC. Since the nominal capacity of the battery cell is 3200 mA, which corresponds to 100% SoC, at 47% SoC, the battery cell capacity would be:
0.47 · 3200 = 1504 mAh ≅ 1500 mAh
Step 2. Read the terminal voltages for 2 selected discharge curves at the calculated discharge capacity of 1500 mAh. We are going to select the 0.2C and 2C discharge curves, for which we can read from the image above the terminal voltages:
U1 = 3.64689 V
U2 = 3.24647 V
Step 3. Calculate the current through the internal resistance for both discharge curves:
I1 = 3200 · 0.2 = 640 mA = 0.64 A
I2 = 3200 · 2 = 6400 mA = 6.4 A
Step 4. Apply equation (1) for the equivalent circuit model for both 0.2C and 2C discharge curves and solve for Ri [Ω].
3.64689 = E – 0.64 · Ri
3.24647 = E – 6.4 · Ri
By subtracting the second equation from the first equation we get:
0.40042 = Ri · (6.4 – 0.64)
which gives the internal resistance:
Ri = 0.06952 Ω = 69.52 mΩ
Conclusion: The internal resistance of the battery cell is approximately 69.52 mΩ at 47% state of charge.
Open circuit voltage calculation
The calculation of the open circuit voltage E [V] is fairly simple, now that we know the value of the internal resistance of the battery cell.
Using the values U1 and I1 for the 0.2C discharge curve, we can write equation (1) as:
3.64689 = E – 0.64 · 0.06952
Solving for E, gives the value of the terminal voltage:
E = 3.64689 + 0.0444928 = 3.6913828 V
when the battery cell is discharged with 640 mA at 47 % state of charge.
Power loss calculation
Having the internal resistance of the battery cell, we can calculate the power loss Ploss [W] for a specific current as:
Ploss = I2 · Ri (eq. 2)
For example, at 47 % SoC, if the output current is 5 A, the power loss of the battery cell would be:
Ploss = 52 · 0.06952 = 1.738 W
Conclusions
The internal resistance of a battery cell can have a significant impact on the performance of an entire battery pack in an electric vehicle (EV). When the internal resistance of a battery cell is high, it can lead to a decrease in the overall capacity of the battery pack, as well as a decrease in the efficiency of the pack. This is because the internal resistance of a battery cell creates a voltage drop within the cell, which can cause the cell to heat up and decrease in performance.
In addition, the internal resistance of a battery cell can also affect the rate at which the cell can charge and discharge, which can impact the overall performance of the battery pack. For example, if the internal resistance of a battery cell is high, it may take longer to charge the pack and the pack may not be able to discharge as quickly as desired.
On the other hand, a battery pack with low internal resistance in its cells will generally have better performance, as it will be able to charge and discharge more quickly and efficiently.
References
[1] Carlos Pastor-Fernández, Energy Storage Systems – Course Notes, The University of Warwick, 2022.
[2] Panasonic NCR18650B lithium-ion battery cell datasheet.




Foo Shen Hwang
Dear Sir/Madam,
Would you be able to send me the article/course notes of Carlos Pastor-Fernández, Energy Storage Systems? I am currently conducting some research on battery technologies and would like to cite this article for my current work.
Regards,
Foo
ElBucaro
For correction, the nominal capacity of the cell is in mAh and not mA. Very useful material, thank you!