Table of Contents
- Air fuel ratio definition
- Air fuel ratio formula
- Air fuel ratio for different fuels
- How stoichiometric air fuel ratio is calculated
- Lambda air fuel ratio
- Air fuel ratio and engine performance
- Air fuel ratio calculator
- Impact of air fuel ratio on engine emissions
- Lambda closed-loop combustion control
Air fuel ratio definition
Thermal engines use fuel and oxygen (from air) to produce energy through combustion. To guarantee the combustion process, certain quantities of fuel and air need to be supplied in the combustion chamber. A complete combustion takes place when all the fuel is burned, in the exhaust gas there will be no quantities of unburnt fuel.
Air fuel ratio is defined as the ratio of air and fuel of a mixture prepared for combustion. For example, if we have a mixture of methane and air which has the air fuel ratio of 17.5, it means that in the mixture we have 17.5 kg of air and 1 kg of methane.
The ideal (theoretical) air fuel ratio, for a complete combustion, is called stoichiometric air fuel ratio. For a gasoline (petrol) engine, the stoichiometric air fuel ratio is around 14.7:1. This means that, in order to burn completely 1 kg of fuel, we need 14.7 kg of air. The combustion is possible even is the AFR is different than stoichiometric. For the combustion process to take place in a gasoline engine, the minimum AFR is around 6:1 and the maximum can go up to 20:1.
When the air fuel ratio is higher than the stoichiometric ratio, the air fuel mixture is called lean. When the air fuel ratio is lower than the stoichiometric ratio, the air fuel mixture is called rich. For example, for a gasoline engine, an AFR of 16.5:1 is lean and 13.7:1 is rich.
Air fuel ratio formula
In the context of internal combustion engines, air fuel ratio (AF or AFR) is defined as the ratio between the mass of air ma and mass fuel mf, used by the engine when running:
\[\bbox[#FFFF9D]{AFR = \frac{m_a}{m_f}} \tag{1}\]The inverse ratio is called fuel-air ratio (FA or FAR) and it’s calculated as:
\[FAR = \frac{m_f}{m_a} = \frac{1}{AFR} \tag{1}\]Air fuel ratio for different fuels
In the table below we can see the stoichiometric air fuel ratio for several fossil fuels.
| Fuel | Chemical formula | AFR |
| Methanol | CH3OH | 6.47:1 |
| Ethanol | C2H5OH | 9:1 |
| Butanol | C4H9OH | 11.2:1 |
| Diesel | C12H23 | 14.5:1 |
| Gasoline | C8H18 | 14.7:1 |
| Propane | C3H8 | 15.67:1 |
| Methane | CH4 | 17.19:1 |
| Hydrogen | H2 | 34.3:1 |
Source: wikipedia.org
For example, in order to burn completely 1 kg of ethanol, we need 9 kg of air and to burn 1 kg of diesel fuel, we need 14.5 kg of air.
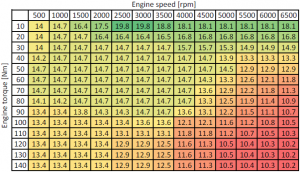
Spark ignition (SI) engines usually run on gasoline (petrol) fuel. The AFR of the SI engines varies within the range 12:1 (rich) to 20:1 (lean), depending on the operating condition of the engine (temperature, speed, load, etc.). Modern internal combustion engines operate as much as possible around the stoichiometric AFR (mainly for gas after-treatment reasons). In the table below you can see an example of a SI engine AFR, function of engine speed and torque.
Compression ignition (CI) engines usually run on diesel fuel. Due to the nature of the combustion process, CI engines always run on lean mixtures, with AFR between 18:1 and 70:1. The main difference, compared with SI engines, is that CI engines run on stratified (non homogeneous) air fuel mixtures, while SI run on homogeneous mixtures (in case of port-injection engines).
The table above is entered in a Scilab script and a contour plot is generated.
EngSpd_rpm_X = [500 1000 1500 2000 2500 3000 3500 4000 4500 5000 5500 6000 6500];
EngTq_Nm_Y = [10;20;30;40;50;60;70;80;90;100;110;120;130;140];
EngAFR_rat_Z = [14 14.7 16.4 17.5 19.8 19.8 18.8 18.1 18.1 18.1 18.1 18.1 18.1;
14 14.7 14.7 16.4 16.4 16.4 16.5 16.8 16.8 16.8 16.8 16.8 16.8;
14 14.7 14.7 14.7 14.7 14.7 14.7 15.7 15.7 15.3 14.9 14.9 14.9;
14.2 14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.7 13.9 13.3 13.3 13.3;
14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.5 12.9 12.9 12.9;
14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.3 13.3 12.6 12.1 11.8;
14.7 14.7 14.7 14.7 14.7 14.7 14.7 14.7 13.6 12.9 12.2 11.8 11.3;
14.1 14.2 14.7 14.7 14.7 14.7 14.7 14.7 13.3 12.5 11.9 11.4 10.9;
13.4 13.4 13.8 14.3 14.3 14.7 14.7 13.6 13.1 12.2 11.5 11.1 10.7;
13.4 13.4 13.4 13.4 13.4 13.6 13.6 12.1 12.1 11.6 11.2 10.8 10.5;
13.4 13.4 13.4 13.4 13.1 13.1 13.1 11.8 11.8 11.2 10.7 10.5 10.3;
13.4 13.4 13.4 13.4 12.9 12.9 12.5 11.6 11.3 10.5 10.4 10.3 10.2;
13.4 13.4 13.4 13.4 12.9 12.9 12.5 11.6 11.3 10.5 10.4 10.3 10.2;
13.4 13.4 13.4 13.4 12.9 12.9 12.5 11.6 11.3 10.5 10.4 10.3 10.2];
contour(EngSpd_rpm_X,EngTq_Nm_Y,EngAFR_rat_Z',30)
xgrid()
xlabel('Engine speed [rpm]')
ylabel('Engine torque [Nm]')
title('x-engineer.org')
Running the Scilab instructions above will generate the following contour plot:
How stoichiometric air fuel ratio is calculated
In order to understand how the stoichiometric air fuel ratio is calculated, we need to look at the combustion process of the fuel. Combustion is basically a chemical reaction (called oxidation) in which a fuel is mixed with oxygen and produces carbon dioxide (CO2), water (H2O) and energy (heat). Take into account that, in order for the oxidation reaction to occur we need an activation energy (spark or high temperature). Also, the net reaction is highly exothermic (with heat release).
\[\text{Fuel}+\text{Oxygen}\xrightarrow[high \text{ } temperature \text{ (CI)}]{spark \text{ (SI)}} \text{Carbon dioxide} + \text{Water} + \text{Energy}\]
Example 1. For a better understanding, let’s look at the oxidation reaction of methane. This is a pretty common chemical reaction, since methane is the primary component of natural gas (in proportion of around 94 %).
Step 1. Write the chemical reaction (oxidation)
\[CH_4 + O_2 \rightarrow CO_2 + H_2O\]Step 2. Balance the equation
\[CH_4 + {\color{Red} 2} \cdot O_2 \rightarrow CO_2 +{\color{Red} 2} \cdot H_2O\]Step 3. Write down the standard atomic weight for each atom
\[ \begin{split}\text{Hydrogen} &= 1.008 \text{ amu}\\
\text{Carbon} &= 12.011 \text{ amu}\\
\text{Oxygen} &= 15.999 \text{ amu}
\end{split} \]
Step 4. Calculate the mass of fuel, which is 1 mol of methane, made up from 1 atom of carbon and 4 atoms of hydrogen.
\[m_f =12.011 + 4 \cdot 1.008 = 16.043 \text{ g}\]Step 5. Calculate the mass of oxygen, which consists of 2 moles, each mol made up from 2 atoms of oxygen.
\[m_o =2 \cdot 15.999 \cdot 2= 63.996 \text{ g}\]Step 6. Calculate the necessary mass of air which contains the calculated mass of oxygen, taking into account that air contains around 21 % oxygen.
\[m_a = \frac{100}{21} \cdot m_o=\frac{100}{21} \cdot 63.996 = 304.743 \text{ g}\]Step 7. Calculate the air fuel ratio using equation (1)
\[AFR = \frac{m_a}{m_f} = \frac{304.743}{16.043} = 18.995 \]The calculated AFR for methane is not exactly as specified in the literature. The difference might come from the fact that, in our example, we made several assumptions (air contains only 21 % oxygen, the products of the combustion are only carbon dioxide and water).
Example 2. The same method can be applied for the combustion of gasoline. Considering that gasoline is made up from iso-octane (C8H18), calculate the stoichiometric air fuel ratio for gasoline.
Step 1. Write the chemical reaction (oxidation)
\[C_{8}H_{18} + O_2 \rightarrow CO_2 + H_2O\]Step 2. Balance the equation
\[C_{8}H_{18} + {\color{Red} {12.5}} \cdot O_2 \rightarrow {\color{Red} 8} \cdot CO_2 +{\color{Red} 9} \cdot H_2O\]Step 3. Write down the standard atomic weight for each atom
\[ \begin{split}\text{Hydrogen} &= 1.008 \text{ amu}\\
\text{Carbon} &= 12.011 \text{ amu}\\
\text{Oxygen} &= 15.999 \text{ amu}
\end{split} \]
Step 4. Calculate the mass of fuel, which is 1 mol of iso-octane, made up from 8 atoms of carbon and 18 atoms of hydrogen.
\[m_f =8 \cdot 12.011 + 18 \cdot 1.008 = 114.232 \text{ g}\]Step 5. Calculate the mass of oxygen, which consists of 12.5 moles, each mol made up from 2 atoms of oxygen.
\[m_o =12.5 \cdot 15.999 \cdot 2= 399.975 \text{ g}\]Step 6. Calculate the necessary mass of air which contains the calculated mass of oxygen, taking into account that air contains around 21 % oxygen.
\[m_a = \frac{100}{21} \cdot m_o=\frac{100}{21} \cdot 399.975 = 1904.643 \text{ g}\]Step 7. Calculate the air fuel ratio using equation (1)
\[AFR = \frac{m_a}{m_f} = \frac{1904.643}{114.232} = 16.673 \]Again, the calculated stoichiometric air fuel ratio for gasoline is slightly different that the one provided in literature. Thus, the result is acceptable since we made a lot of assumptions (gasoline contains only iso-octane, air contains only oxygen in proportion of 21 %, the only products of combustion are carbon dioxide and water, the combustion is ideal).
Lambda air fuel ratio
We have seen what is and how to calculate the stoichiometric (ideal) air fuel ratio. In reality, internal combustion engines do not work exactly with ideal AFR, but with values close to it. Therefore we’ll have an ideal and a actual air fuel AFR ratio. The ratio between the actual air fuel ratio (AFRactual) and the ideal/stoichiometric air fuel ratio (AFRideal) is called equivalence air fuel ratio or lambda (λ).
\[\bbox[#FFFF9D]{\lambda = \frac{AFR_{actual}}{AFR_{ideal}}} \tag{3}\]For example, the ideal air fuel ratio for a gasoline (petrol) engine is 14.7:1. If the actual/real AFR is 13.5, the equivalence factor lambda will be:
\[\lambda = \frac{13.5}{14.7} = 0.92\]Depending on the value of lambda, the engine is told to work with lean, stoichiometric or rich air fuel mixture.
| Equivalence factor | Air fuel mixture type | Description |
| λ < 1.00 | Rich | There is not enough air to burn completely the amount of fuel; after combustion there is unburnt fuel in the exhaust gases |
| λ = 1.00 | Stoichiometric (ideal) | The mass of air is exact for a complete combustion of the fuel; after combustion there is no excess oxygen in the exhaust and no unburnt fuel |
| λ > 1.00 | Lean | There is more oxygen than required to burn completely the amount of fuel; after combustion there is excess oxygen in the exhaust gases |
Depending on the type of fuel (gasoline or diesel) and the type of injection (direct or indirect), an internal combustion engine can function with lean, stoichiometric or rich air fuel mixtures.
For example, the Ford Ecoboost 3-cylinder engine runs with stoichiometric air fuel ratio for idle to medium engine speed and complete load range, and with rich air fuel mixture at high speed and load. The reason for which it runs with rich mixture at high engine speed and load is engine cooling. The additional fuel (which will remain unburnt) is injected to absorb heat (through evaporation), reducing this way the temperature in the combustion chamber.
A compression ignition (diesel) engine runs all the time with lean air fuel mixture, the value of the equivalence factor (λ) depending on the engine’s operating point (speed and torque). The reason for this is the working principle of a diesel engine: controlling load not through air mass (which is always in excess) but through fuel mass (injection time).
Remember that a stoichiometric equivalence factor (λ = 1.00) means an air fuel ratio of 14.7:1 for gasoline engines and 14.5:1 for diesel engines.
Air fuel ratio and engine performance
The engine performance in terms of power and fuel consumption is highly dependent on the air fuel ratio. For a gasoline engine, the lowest fuel consumption is obtained at lean AFR. The main reason is that there is enough oxygen available to burn completely all the fuel which translates in mechanical work. On the other hand, the maximum power is obtained with rich air fuel mixtures. As explained before, putting more fuel in the cylinder at high engine load and speed, cools down the combustion chamber (through fuel evaporation and heat absorption) which allows the engine to produce maximum engine torque thus maximum power.
In the figure above we can see that we can not get the maximum power of the engine and the lowest fuel consumption with the same air fuel ratio. The lowest fuel consumption (best fuel economy) is obtained with lean air fuel mixtures, with an AFR of 15.4:1 and an equivalence factor (λ) of 1.05. The maximum engine power is produced with rich air fuel mixtures, with an AFR of 12.6:1 and an equivalence factor (λ) of 0.86. With a stoichiometric air fuel mixture (λ = 1), there is a compromise between maximum engine power and minimum fuel consumption.
Compression ignition (diesel) engines always run on lean air fuel mixtures (λ > 1.00). Most of the modern diesel engines run with λ between 1.65 and 1.10. The maximum efficiency (lowest fuel consumption) is obtained around λ = 1.65. Increasing the fuel amount above this value (going towards 1.10) will produce more soot (unburnt fuel particles).
There is an interesting study performed by R. Douglas on 2-stroke cycle engines. In his doctoral thesis “Closed Cycle Studies of a Two-Stroke Cycle Engine“, R. Douglas comes with a mathematical expression of the combustion efficiency (ηλ) function of equivalence factor (λ).
For spark ignition (gasoline engine) with an equivalence factor between 0.80 and 1.20, the combustion efficiency is:
\[\eta_{\lambda}=-1.6082+4.6509 \cdot \lambda – 2.0746 \cdot \lambda^2 \tag{4}\]For compression ignition (diesel engine) with an equivalence factor between 1.00 and 2.00, the combustion efficiency is:
\[\eta_{\lambda}=-4.18+8.87 \cdot \lambda – 5.14 \cdot \lambda^2 + \lambda^3 \tag{5}\]For diesel engines, if the equivalence factor goes above 2.00, the combustion efficiency is maximum (1.00 or 100 %).
We can use a Scilab script to plot the variation of the combustion efficiency function of the equivalence factor.
lmbd_g = [0.80:0.01:1.20];
lmbd_d = [1.00:0.01:2.00];
eff_lmbd_g = -1.6082+4.6509*lmbd_g-2.0746*lmbd_g.^2;
eff_lmbd_d = -4.18+8.87*lmbd_d-5.14*lmbd_d.^2+lmbd_d.^3;
plot(lmbd_g,eff_lmbd_g,'b','LineWidth',2)
hold
plot(lmbd_d,eff_lmbd_d,'r','LineWidth',2)
xgrid()
xlabel('$\lambda \text{ [-]}$')
ylabel('$\eta_{\lambda} \text{ [-]}$')
title('x-engineer.org')
legend('gasoline','diesel',4)
Running the Scilab instructions above outputs the following graphical window.
As you can see, the compression ignition (diesel) engine, at stoichiometric air fuel ratio has a very low combustion efficiency. The best combustion efficiency is obtained at λ = 2.00 for diesel and λ = 1.12 for spark ignition (gasoline) engines.
Air fuel ratio calculator
| ma [g] | Fuel type | λ [-] |
| mf [g] | ηλ [%] |
Observation: The combustion efficiency is only calculated for diesel and gasoline (petrol) fuel, using equations (4) and (5). For the other fuels, the combustion efficiency calculation is not available (NA).
Impact of air fuel ratio on engine emissions
Internal combustion engine exhaust gas emissions depend heavily on the air fuel ratio (equivalence factor). The main exhaust gas emissions in ICE are summarised in the table below.
| Exhaust gas emission | Description |
| CO | carbon monoxide |
| HC | hydrocarbon |
| NOx | nitrogen oxides |
| Soot | unburnt fuel particles |
For a gasoline engine, CO, HC and NOx exhaust gas emissions are heavily influenced by air fuel ratio. CO and HC are mainly produced with rich air fuel mixture, while NOx with lean mixtures. So, there in no fixed air fuel mixture for which we can obtain the minimum for all exhaust emissions.
A three way catalyst (TWC), used for gasoline engines, has the highest efficiency when the engine operates in a narrow band around stoichiometric air fuel ratio. The TWC converts between 50 … 90 % of hydrocarbons and 90 … 99 % of carbon monoxide and nitrogen oxides, when the engine runs with λ = 1.00.
Lambda closed-loop combustion control
In order to meet the exhaust gas emissions regulations, it is critical for internal combustion engines (especially gasoline) to have an accurate control of the air fuel ratio. Therefore, all of the modern internal combustion engines have closed-loop control for air fuel ratio (lambda).
- air mass flow sensor
- primary catalyst
- secondary catalyst
- fuel injector
- upstream lambda (oxygen) sensor
- downstream lambda (oxygen) sensor
- fuel supply circuit
- intake manifold
- exhaust manifold
The critical component for the system to work is the lambda (oxygen) sensor. This sensor measures the level of oxygen molecules in the exhaust gas and sends the information to the engine electronic control unit (ECU). Based on the value of the oxygen sensor reading, the gasoline engine ECU will adjusts the level of fuel mass in order to keep the air fuel ratio around the stoichiometric level (λ = 1.00).
For example (gasoline engines), if the level of oxygen molecules is above the threshold for stoichiometric level (therefore we have a lean mixture), at the next injection cycle, the injected fuel amount will be increased in order to make use of the excess air. Bear in mind that the engine will always transition from lean mixture to rich mixture between injection cycles, which will give an “average” of stoichiometric air fuel mixtures/ratio.
For diesel engines, since it always runs on lean air fuel ratio, lambda control is performed in a different manner. The end goal being still the same, control of the exhaust gas emissions.
For any questions or observations regarding this tutorial please use the comment form below.
Don’t forget to Like, Share and Subscribe!








naman
can you please show plots of diesel emissions formation w.r.t. AFR?
Ricardo Calles
One important factor concerning the “Catalyst Efficiency Function on Air Fuel Ratio” that is misleading is that excess NOx production only occurs when combustion temps exceed 2370 degrees F. In fact, properly designed lean burn engines actually run cooler due to less fuel being burnt along with more air to cool the combustion chamber.
David
Your article is enjoyable, but may cause confusion in understanding due to a lack of descriptive tuning goals. The use of any Lambda is based on the efficiency goals of the tune. Maximum torque, maximum economy, minimum exhaust emissions (of specific types), etc. For example, your graphic “Stoichiometric (Ideal)” is missing an important goal of “Best emissions” at the center, and while being neither best for power or economy, is the common use of stoichiometric Lambda. This would help clarify statements such as “The reason for which it runs with rich mixture at high engine speed and load is engine cooling.” is not accurate, and the use of stoich at cruise is to address emissions, while rich mixtures are primarily for torque (with side-effects such as cooling and burn rate). From the goals, we can see that if excluding emissions goals, fueling would typically be only lean for economy or rich for torque, almost never using the stoichiometric value except in-transition. I hope that helps perspective and “connecting the dots”.
Icaro Vilasboas
Excellent article. I am very interested in your text references.
Dakhil Al-shehab
I have Porsche Cayman R 2012 tune it to get more power with full straight away exhaust” headers & cutback” so lambda when I stop and the rpm around (700 ) show me 0.995 when I make the car under loud 6000 rpm its jump to 1.05 or around.. and I have check engine sign. are this situation are ok ?!
Juan 12
Hello everyone,
I have some mixtures of etanol with gasoline in proportions of percenties E10,E20,E50 and E85. I want to know how I do to obtain stoichiometric mixtures of them? On chemical terms. Thanks
Rhett
Am I misinterpreting the contour plot? There seems to be missing (and extra) plot data based on the engine speed/engine torque/AF chart. For example, for 500 RPM, there appears to be only 6 data points, and most of those are at engine torque values not indicated in your Scilab script (and colored chart).
julien thibodeau
very good tutorial. just one small positive criticism. gasoline engines can use oxygen sensors or lambda sensors, they do the same job but aren’t the same. oxygen sensors are called narrow band sensors, they work well around stoich but cannot read very lean or rich. lambda sensors are called wide band sensors, they are designed to read very rich or very lean. example, during full throttle operation, you ecu uses whats called p.e. or power enrichment, its what makes the afr fall to ex. 12.5:1 . an oxygen sensor will just read rich or generate 0.9 volts flat. wide band sensors can still read this afr and relay precise information to the ecu. be careful when calling an oxygen sensor a lambda sensor because though they perform a similar task they have a few differences.
glenn
I have an issue with very high fuel consumption. 2L superchargered 8 valve 4 cylinder. A little cammy, AFR of 14.00 at idle, (any less and it wont idle smoothly) and 14.5 crusing, 12:0 when on boost (only 5psi) car drives well but uses heeaps of fuel. Oil smells like raw fuel also. The injectors are questionable. I was thinking would a poor spray pattern consume excess fuel while AFR’s still remain OK around 14.5/15 cruisng @ 100km/.h. Fuel consumption is something like 20L / 100km which is crazy. Raw fuel un-atomized won’t burn, or combine with oxygen and thus goes straight out the exhaust or washers down the cylinder walls?
Lily
I’d like to cite this text can I get an author name, and/or year of publication?
Piyush
Can I get a source for the combustion efficiency of Diesel and Gasoline plot above
Bruno Chaves
Hi, I see a lot about the hydrogen ration and people represent Hydrogen as H2, H2 is DiHydrogen
So is the stoichiometric ratio here for Dihydrogen, or is it for hydrogen?
Klam
For the calculation of AFR you are using the volumetric percentage of oxygen in the air (21%) instead of the mass percentage (23.3%). Fix it so the numbers are closer to the literature values.
Grant
Great article, better than so-called car mags that are light on facts. I have a question – what would water getting into the exhaust do to lambda? I have a turbo car and get erratic/lean readings and I can’t find an air leak.
AN
Pretty sure the ECU would treat that as it would a vac leak and add fuel/increase injector pulse width? This is a great article and discussion even for self-professed gear heads like myself.
Allen lougheed
I’m running a turbo charged engine using 94 octane chevron gas running 9 pounds of boost inline ford with the Cleveland head on it,what would you think The boost limit would be with that fuel being used that makes 702hp @6300
Filiph Sandberg
Great summary! I would like to see some sources from where you gathered this information so I can use this in my work.
TST
In the isooctane example, you’ve implicitly assumed the molecular weight of air to be the same as that of O2. Ideally, you would need 12.5*(32 + 3.76(28)) g of air (or simply 12.5*4.76*28.84 g of air) for 114 g (i.e. 1 mole) of IC8H18. That would bring down the ratio to 15.05.
David Atwood
I have figured out why you’re answer in the example 1 was wrong. You have assumed a ratio of Oxygen to Nitrogen of 21% per 100%, which is only true for a volumetric percentage. In that step you have converted to grams, so you should have used the percentage of weight. In fact, oxygen constitutes 23.14% of the air by weight. So if the fraction were 23.14/100, then the MAir=(100/23.14)*MOxygen=4.322*63.996=276.56 grams. This yields an Air to Fuel Ratio by weight of 17.24. Much closer to the chart.
Fahmi ali
Very usefull that presentation for my job..thanks a lot
Abderrahman MAHIDDINI
In the litterature, if found that the NOx concentration in the exhaust rise when the equivalence ration is in reach case. but in the present article i see that is quite opposite. So which is the right trend?
Bruno Chaves
So NOx concentrations come from two places:
Nitrate particulates in air, and some additives in the fuel, but in the fuel they are negligible.
Most of them actually com from the air that the intake sucks in for combustion.
You can do the experiment:
Run your vehicle in a city and measure nox emissions, then go to the country and measure nox emissions, there should be a substantial difference. by country i mean forest, not near farming fields, a lot of nitrates around.
davESKI
WHAT WILL HAPPEN TO THE LAMDA VALUE IF A GAS ENGINE HAS TOO MUCH COMPRESSION? MY EXAMPLE, I HAVE A CAR WITH HIGH COMPRESSION AND THE COMPUTER IS ADDING FUEL THINKING IT IS LEAN….IS THAT “LEAN” CONDITION DUE TO TOO HIGH COMPRESSION “+25%”
Jon Snow
I want to cite this, can you state your name & year of publication
Shakaib
Excellent. Are there any other articles on other engin parameters?
Stelios
Excellent tutorial!
Wonder if there is an equivalent for marine engines (does size make a difference?) and if a third fuel could be added in the tutorial arguments: natural gas (methane).
Alan Rosean
15.4:1 for best fuel economy. Just the number I have been looking for. I am a practical, seat of the pants, mechanic attempting to tune the primary of a 4-bbl carb to improve the fuel economy of a Ford 460; the secondary will operate rich enough for the needed power. Far too many ‘experts’ are saying too lean will damage an engine but what is too lean? If you cannot measure it then you cannot troubleshoot it, all you can do is guess. The engine I built is super strong and under powered at 300+ hp so better cruise fuel economy would be nice. I am monitoring the air/fuel mixture and jetting as needed. Thank you for the article.
David
Don’t misread the example. That example is of a specific engine, fuel, load, drag, etc. Also note the ignition timing to extract the energy is a critical factor. My last mileage testing showed a GM V8 producing peak economy at 16.5-16.7 AFR on E10 pump gas (Lambda ±1.12), but timing was tuned for minimizing pumping losses and exhaust heat.
EDUARDO
Do you have information about EGR mix. impact in lambda result? specially on ICE
nicky
How about plotting a graph of the stoichiometric air fuel ratio for petrol mixed with ethanol from 10-30% showing how the varying percentage of ethanol changes the stoichiometric A/F/R compared to the other percentages & ethanol free petrol?
mallick
Excellent U done have good job.
AI
You’ve made an error in the calculation of AFR for methane in example 1. You’ve used 63.996 as the mass for fuel instead of 16.043.
Anthony Stark
Hi,
Thanks for noticing, I’ve corrected it.
Christopher Simmins
Excellent graphs descriptions etc …however in last image the word ‘motor’ should be replaced with ‘engine’.
jonathan
disagree
Corey A
That would depend largely on what century you lived in, if you are living in the present century, I’d say either would mean almost the same thing. You can look that up on any search “engine”…
David
Interesting catch of a word differentiation coming back into vogue, e.g., in a hybrid car, using motor (electric) and engine (combustion) greatly simplifies and avoids confusion. The world isn’t done with different definitions just yet…