All objects surrounding us are made from matter. The universe is also containing matter but at a different scale. Matter can exist in several states (also called phases). The most common, fundamental states of matter are:
- gas
- liquid
- solid
There is also a fourth state, quite common as well, but less intuitive:
- plasma
The state in which an object is depends on two physical attributes: pressure and temperature. Let us take as example water. On Earth, the most common state for water is liquid. This is because the global average annual temperature is 15 °C and the atmospheric pressure has 1 bar. In these conditions water will be in liquid state.
The higher the temperature (heat, enthalpy) of matter, the higher the kinetic energy of the molecules. This will cause the molecules to vibrate at higher amplitudes and frequencies, which will break the inter-molecular forces and set the molecules apart from each other. That is why, in general, the higher the temperature, the lower the density.
Depending on temperature (assuming constant pressure), matter can be found in solid, liquid or gas state.
SOLIDS
Matter in solid state have the highest density, with the molecules tightly packed together. Compared to other states, the kinetic energy in a solid is low. Solids have a definite shape and volume, if placed in a container, they don’t conform to the geometry of the container. Having a definite volume, even if compressed at high pressure will not compress them in a smaller volume, solids are not compressible.
A solid can become a liquid, the process being called melting or fusion. For example, ice (solid) at temperatures above 0 °C melts and becomes liquid water. The same process applies also to iron, which becomes liquid at temperatures above 1500 ºC.
Solids can also transition directly to gases, the process being called sublimation. Naphthalene, the main ingredient of traditional mothballs, is an organic compound with formula C10H8. It is a solid that sublimes at standard atmospheric temperature and low temperatures.
LIQUIDS
Liquids have lower density than solids (except water) but higher kinetic energy. They also have a definite volume so they can’t be compressed. The molecules of water are more loose, compared with solids, and they can move relative to each other. A liquid doesn’t have a definite shape and, if place in a container, takes its shape.
A liquid can transition in a solid through a process called solidification (or crystallization). Water, if cooled down below 0 °C, becomes a solid (ice).
The process of transformation from a liquid into a gas is called vaporization. It the case of water, if heated up to 100 °C, it starts to boil and transforms into steam.
GASES
Compared to the other states of matter, gases have the lowest density and the highest kinetic energy. The molecules within a gas have a lot of space between themselves and, if not contained, will spread out indefinitely. If placed in a container, the gas occupies the whole volume. Put under pressure, the spaces between the molecules will get smaller and the volume of the gas will decrease. A gas is compressible.
A gas can be transformed into a liquid through a process called condensation. If a gas is cooled down enough, the kinetic energy of the molecule will not be capable anymore to overcome the inter-molecular forces. This will result in an accumulation of molecules which will form the liquid. For example, water vapors (steam) if cooled down below 100 °C will start to condensate into liquid water.
The transformation of a gas directly into a solid is called deposition. If a gas is placed directly in a very cold environment, it will transform in tiny solid particles. For example, water vapors transforms into ice crystals when the temperatures are below 0 °C.
PLASMA
Plasma is to most common state of matter in the universe but not very common on Earth. Given a very high amount of heat, a gas can be transformed into plasma. In a plasma state, the atoms are ionized, which separates the electrons (negative charge) form the ions (positive charge).
Examples of plasma in nature are: lightning, neon light and electrical sparks. Plasma can be also artificially produced, the most common technology being the application of an electric current across a dielectric gas or fluid.
Plasma can only be produced from gases and the reverse process is only from plasma to gas, through deionization.
PHASE DIAGRAMS
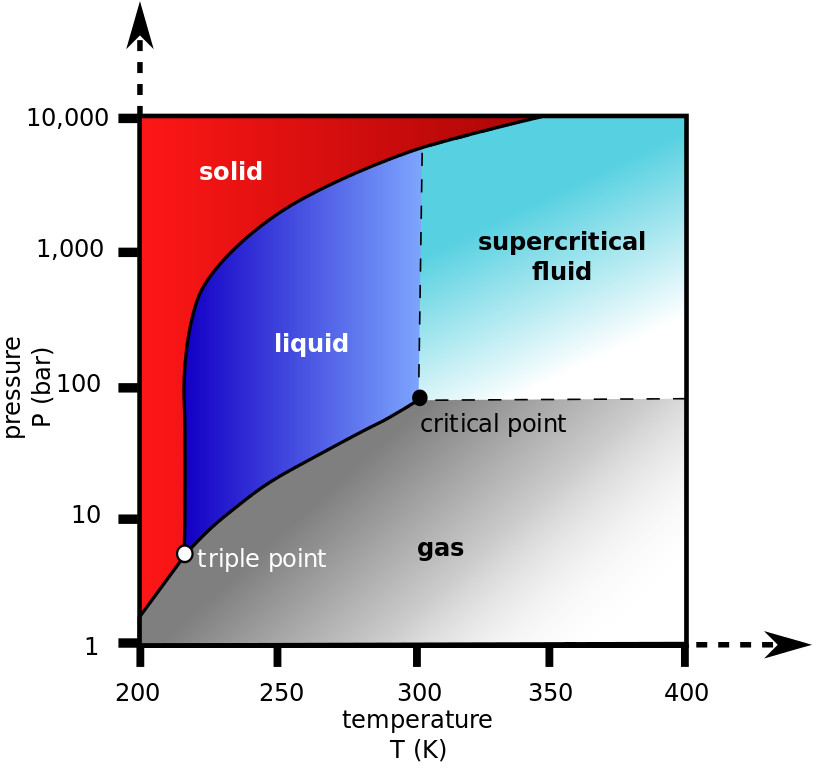
The transformation of matter are best described in phase diagrams. These diagrams show the equilibrium phases within a substance function of temperature and pressure or composition.
Simple phase diagrams (2-D), that can be easily understood, are the pressure–temperature diagrams of a single simple substance, water for example. The vertical axis correspond to pressure and the horizontal one to temperature. The phase diagram shows, in pressure–temperature space, the lines of equilibrium or phase boundaries between the three phases of solid, liquid, and gas.
The triple point is represented by the value of temperature (Ttp) and pressure (ptp) at which all three phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium. For water, the coordinates of the triple point are: 0.01 °C and 0.0061166 bar. At this point, with small changes in pressure and temperature, it’s possible to transform all of the substance into ice, water, or vapor.
The critical point defines the end of the liquid-vapor boundary. It is defined by the critical temperature (Tcr) and critical pressure (pcr). At the critical point the phase boundary vanishes, a liquid and its vapor can coexist in the same state. The critical point for water occurs at 374 °C and 220.888 bar. Near the critical point, water becomes compressible, expandable, a poor dielectric, and a bad solvent for electrolytes. Above the critical point water becomes a supercritical liquid. A substance in this state combines the properties of both liquid and gases and, by fine tuning of the temperature and pressure, it is possible to control the behaviour to be more liquid-like or gas-like.
Water (H2O) and carbon dioxide (CO2) are the most commonly used supercritical fluids.
A particular state of matter is the Bose-Einstein condensates (BEC). By cooling down a substance at extremely low temperatures (close to absolute zero, 0 K = -273.15 °C) molecular vibration comes very close to a complete stop. All the atoms of the substance are gathering together, because there is no kinetic energy to set them apart, creating a “super atom”. BEC substances are mainly used to simulate conditions that might apply in black holes.
For any questions or observations regarding this tutorial please use the comment form below.
Don’t forget to Like, Share and Subscribe!