By definition, internal combustion engines (ICE) burn a mixture of fuel and air (oxygen), in an appropriate ratio, in order to produce mechanical power. In a four-stroke internal combustion engine, the combustion process occurs after the mixture of fuel-air has been induced into the cylinder, properly compressed and a spark generated (in case of a gasoline/petrol fuel).
The start of combustion process is linked to the position of the piston in the cylinder and usually happens before the piston hits the Top Dead Centre (TDC). As a result of the combustion process, the pressure generated in the cylinder rises and its transmitted as a torque to the crankshaft through the piston and connecting rod.
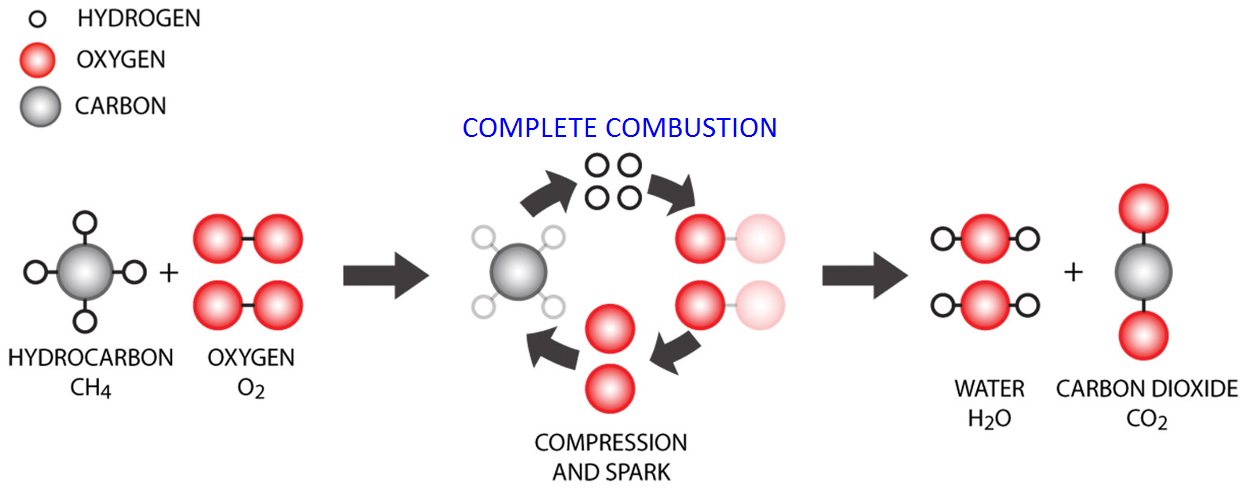
Combustion can be defined as a rapid oxidation process of a fuel, which generates heat and light. With other words, combustion converts the energy stored in the chemical bonds of the fuel into heat. In an engine, combustion occurs as a flame that propagates in the cylinder. A flame (fire) occurs if three components are present: fuel, oxidizing agent (oxigen) and heat. Depending on the type of the fuel, the heat for the occurrence of the flame comes from: a spark (gasoline/petrol engines) or heated (compressed) air (diesel engine).
The main product of an internal combustion engine is mechanical power, this is why we use them as propulsion systems. Together with mechanical power, as a by-product, we also get exhaust gases. Ideally, if the combustion process is complete, the exhaust gases should only be carbon dioxide (CO2) and water vapour (H2O). In reality, mainly due to incomplete combustion, the exhaust gases also contain pollutant emissions: oxides of nitrogen (NOx), unburnt hydrocarbons (HC), carbon monoxide (CO), soots/particles (PM), polyaromatics, aldehydes ketones and nitro-olefins.
Attention: Carbon dioxide (CO2) is not a pollutant emission because it is not harmful to humans or environment. The problem with CO2 is that it’s a green-house effect gas and contributes to global warming.
If we take methane (CH4) as fuel example, after its complete combustion process, ideally, will get only water (H2O) and carbon dioxide (CO2).
In reality the fuel is mixed with air and not pure oxygen (O2). Since more than 70 % percent of the air is nitrogen (N2), a part from it will react with oxygen and form oxides of nitrogen (NOx). Also, since the air-fuel mixture is not 100% homogeneous, a part of the fuel will not burn (oxidize), which means that in the exhaust gases we’ll have unburnt hidrocarbons (HC). For the same reason, the oxigen may also be insufficient for combustion, which will generate carbon monoxide (CO).
Due to the limitations of the fuel conversion efficiency, there are unintended outputs of an internal combustion engine. The limitations of the fuel conversion efficiency are caused by:
- inconsistent cylinder pressure
- inconsistent fuel mass
- excessive peak cylinder pressure
- excessive/insufficient oxygen in the cylinder
- excessive exhaust gas pressure
In the table below there is a summary of the intended outputs (what we want) of an internal combustion engine and the real outputs (what we actually get).
Inputs:
| Combustion process (oxidation) | Intended outputs:
|
Real outputs (additional to the intended outputs):
|
The efficiency and power output of an internal combustion engine depends on a great extent on the pressure characteristic of the engine (p-V diagram), which depends on the engine cycle (4-stroke): induction/intake, compression, power (combustion) and exhaust.
The generation of pollutant exhaust gas emissions also depends on a great extent on the sequence of engine phases (engine cycle). From this point of view, there are differences between diesel and gasoline (petrol), especially regarding how the air-fuel mixture is created and how the combustion process is triggered. Also, the properties of the fuel determines how the combustion is initiated, how the flame is propagated and also the content of the exhaust gas. In the table below we can see some of the main properties of gasoline (petrol) and diesel fuel.
| Gasoline (Petrol) | Diesel | |
| Density [kg/L] | 0.745 | 0.832 |
| Heating value [MJ/kg] | 43.2 | 43.1 |
| Volumetric energy [MJ/L] | 32.18 | 35.86 |
| *Flash point [° C] | -43 | 52 |
| *Auto-ignition point [° C] | 280 | 256 |
| Number of C atoms per molecule | 4 – 12 | 9 – 25 |
| CO2 emissons [kg/kg of fuel] | 2.30 | 2.65 |
| CO2 emissions [g/MJ] | 73.38 | 73.25 |
| Engine efficiency [%] | 30 | 40 |
*Flash point definition: the lowest temperature at which the fuel can form an ignitable mixture in air
*Auto-ignition point definition: the lowest temperature at which a gas or vapour will spontaneously ignite without a distinct (localised) ignition source
From the fuel properties table we can draw some conclusions:
- the energy density of diesel is higher than gasoline (petrol); this means that for the same volume of fuel, we get more energy from a diesel engine
- gasoline (petrol) is very volatile, the flash point is much lower than diesel, and can be easily ignited from an external source (spark)
- the CO2 emissions per kg of fuel, for an ideal combustion scenario, is higher for diesel fuel since the number of carbon atoms is also higher; however if we relate the CO2 emissions to the energy value of the fuel, the diesel fuel has slightly lower emissions; also, taking into account the overall engine efficiency of the diesel engines, the CO2 emissions of diesel vehicles are lower than those of gasoline (petrol) vehicles
In the diagram below we can observe the correlation between the piston stroke and combustion phases, for a 4-stroke internal combustion engine, for both diesel (compression ignition) and gasoline/petrol (spark ignition) engines.

Image: Internal combustion engine process – diesel vs. gasoline (petrol)
TDC – Top Dead Center, BDC – Bottom Dead Center
In a gasoline (petrol) internal combustion engine, before the spark is generated, the air-fuel mixture must be homogeneous. This is critical for the flame to appear around the spark and propagate into the cylinder, for a quasi-complete combustion. The electric arc (plasma) generated by the spark plug reaches temperatures of 10000 °C and triggers the combustion process. The air motion is also important since the higher the turbulence into the cylinder, the faster the combustion and flame propagation. In a normal combustion process, the speed of the flame into the cylinder, during combustion, is between 25 – 50 m/s.
In a diesel internal combustion engine, the air-fuel mixture is not homogeneous but stratified. Compared with a gasoline/petrol engine, the time allowed for the fuel to mix with air is much shorter, between 5-8 times. If for the gasoline engine the air and fuel are mixed for nearly two piston strokes (intake and compression), for a diesel engine the mixture forms in around 40 – 80 ° of crankshaft rotation. The flame is generated when the fuel is vaporised (during injection) and comes into contact with the hot compressed air.
From the combustion process point of view, we can summarise the differences between a diesel and a gasoline/petrol engine as:
| Diesel | Gasoline/Petrol | |
| Combustion trigger | Internal (high temperature of compressed air) | External (spark plug) |
| Air-fuel mixture | Locally stratified, lean in the cylinder | Homogeneous (lean, stoichiometric or rich) |
| Load control | Qualitative (through fuel mass control), unthrottled | Quantitative (through air mass control), throttled |
| Speed of combustion | Slow, which leads to low engine speed but high torque | Fast, which leads to high engine speed and power |
| Pollutant emissions | High soot and NOx, low CO and HC | High HC and CO, low soot and NOx |



Khairi
Good write up.
Thank you
pilot
Typo
” For the same reason, the oxigen may also be insufficient”
It’s oxygen not oxigen.
Michael Zagaal
Great illustrations however it focuses mostly on the thermodynamic properties and not quite so much on on chemistry. It would be helpful to explain how typical engines produce more CO2 emissions than they do in liquid fuel.