Internal combustion engines produce mechanical work (power) by burning fuel. During the combustion process the fuel is oxidized (burned). This thermodynamic process releases heat which is transformed partly in mechanical energy.
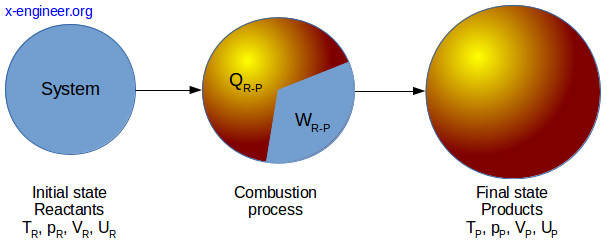
Let’s regard the internal combustion engine as a system, with a defined boundary. In its initial state, the engine will contain some reactants, mainly fuel and air. After the combustion process, the engine will be in a final state, containing combustion products (exhaust gases).
Applying the first law of thermodynamics to our engine system, between the initial and final state, gives:
\[Q_{R-P} – W_{R-P}=U_P – U_R \tag{1}\]where:
Q [J] – heat transfer
W [J] – mechanical work
U [J] – internal energy
T [K] – temperature
p [Pa] – pressure
V [m3] – volume
Combustion efficiency
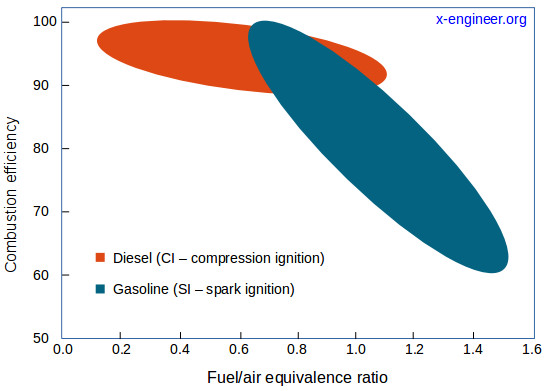
In real engine applications, the combustion process is incomplete. This means that not all the energy content of the fuel supplied to the engine is released through the combustion process. There are several factors which can influence the combustion process, the most important being the fuel-air intake and fuel atomization (size of droplets).
The fuel inside the cylinder needs air (oxygen) to burn. If there is not enough oxygen available, not all the fuel is burnt, therefore only a partial energy is released from combustion (e.g. around 96 %).
If we analyze the exhaust gas of an internal combustion engine, we can see that it contains both incomplete combustion products (carbon monoxide CO, nitrogen oxides NOx, unburnt hydrocarbons HC, soot PM) and complete combustion products (carbon dioxide CO2 and water H2O).
If the engine is running under lean operating conditions the amounts of incomplete combustion products are small, because there is oxygen in excess. Under rich operating conditions these amounts become more substantial since there is insufficient oxygen to complete the fuel combustion.
Because a fraction of the fuel’s chemical energy is not fully released inside the engine during the combustion process, it is useful to define a combustion efficiency.
The combustion efficiency ηc [-] is defined as the ratio between the energy released by the burnt fuel and the theoretical energy content of the fuel mass during one complete engine cycle.
\[\eta_c = \frac{H_R (T_A) – H_P (T_A)}{m_f \cdot Q_{HV}} \tag{2}\]where:
HR [J] – enthalpy (internal energy) of the reactant
HP [J] – enthalpy (internal energy) of the product
TA [K] – ambient temperature
mf [kg] – mass of fuel inducted per cycle
QHV [J/kg] – heating value of the fuel
Heating value
The heating value (also knows as energy value or calorific value) of a fixed quantity of fuel, is the amount of heat released during its combustion. The heating value of a fuel is the magnitude of the heat of reaction measured at constant pressure/volume and standard temperature (26 °C) for the complete combustion of a unit of mass of fuel.
Any fuel has two types of heating values:
- higher heating value (HHV), also know as gross heating values
- lower heating value (LHV), also known as net heating value (is determined by subtracting the heat of vaporization of the water from the higher heating value)
As example, in the table below, we can see the heating values for the most common and alternative fuels used in internal combustion engines:
| Fuel | Lower heating value [MJ/kg] | Higher heating value [MJ/kg] |
| Hydrogen | 119.96 | 141.88 |
| Natural gas | 47.13 | 52.21 |
| Conventional gasoline | 43.44 | 46.52 |
| Conventional diesel | 42.78 | 45.76 |
| Ethanol | 26.95 | 29.84 |
| Liquefied Petroleum Gas (LPG) | 46.60 | 50.14 |
| Liquefied Natural Gas (LNG) | 48.62 | 55.19 |
| Butane | 45.27 | 49.20 |
| Propane | 46.28 | 50.22 |
Thermal conversion efficiency
Thermal conversion efficiency relates the actual work per cycle to the amount of fuel chemical energy released in the combustion process.
Thermal conversion efficiency is defined as the ratio between the work per cycle Wc [J] and the energy released by the burnt fuel.
\[\eta_t = \frac{W_c}{H_R (T_A) – H_P (T_A)} \tag{3}\]The thermal conversion efficiency shows how much of the burnt fuel is converted into useful mechanical work.
Fuel conversion efficiency
The fuel conversion efficiency is defined as the ratio between the useful mechanical work produced by the engine and the theoretical energy content of the fuel mass.
\[\eta_f = \frac{W_c}{m_f \cdot Q_{HV}} \tag{4}\]The work per cycle Wc [J] can be written function of engine power and speed:
\[W_c = \frac{P \cdot n_R}{N} \tag{5}\]where:
P [W] – engine power (indicated)
N [rot/s] – engine speed
nR [-] – number of crankshaft revolutions for each power stroke per cylinder
The fuel mass used per engine cycle mf [kg] can be written function of fuel mass flow rate and engine speed:
\[m_f = \frac{\dot{m}_f \cdot n_R}{N} \tag{6}\]where mf (dot) [kg/s] is the fuel mass flow rate.
Replacing (5) and (6) into (4), gives the expression of the fuel conversion efficiency function of engine power, fuel mass flow rate and fuel heating value:
\[\eta_f = \frac{P}{\dot{m}_f \cdot Q_{HV}} \tag{7}\]Engine specific fuel consumption SFC [kg/J] is the ratio between fuel mass flow rate and indicated engine power:
\[SFC = \frac{\dot{m}_f}{P} \tag{8}\]Replacing (8) in (7) gives the expression of the fuel conversion efficiency function of specific fuel consumption and fuel heating value:
\[\eta_f = \frac{1}{\text{SFC} \cdot Q_{HV}} \tag{9}\]The fuel conversion efficiency is also the product between the combustion efficiency and the thermal conversion efficiency.
\[\eta_f = \eta_c \cdot \eta_t \tag{10}\]How to calculate fuel conversion efficiency
A gasoline engine at a steady engine speed has a fuel mass flow rate of 6 g/s and outputs 80 kW of power. Calculate the fuel conversion efficiency using the lower heating value of conventional gasoline.
\[\eta_f = \frac{P}{\dot{m}_f \cdot Q_{HV}} = \frac{80 \cdot 10^3}{6 \cdot 10^{-3} \cdot 43.44 \cdot 10^6 } = 0.307\]The fuel conversion efficiency of the engine is 30.7 %.
Don’t forget to Like, Share and Subscribe!

Chris
Can you recommend any books specific to internal combustion engine BSFC and fuel efficiency?
Or any engineering books about ICE’s?
Sambo
Why aren’t your equations displayed?
Anthony Stark
Sometime the Mathjax module is not properly loaded. Unfortunately I can’t do much about it since it’s between the client (browser) and the Mathjax repository. Try reloading (F5) the web page and it should display correctly.
B Sai Varma
I want to calculate combustion efficiency only by using fuel parameters(i.e without using Torque or Power). Is that possible